Most articles about FES cycling focus on the obvious: bigger leg muscles, better fitness, preserved bone density. These are real, and they are worth talking about. But in my experience, they should not be the only reason people who stick with FES cycling for ten or twenty years do so. The real reason is what does not happen.
What does not happen is the third UTI of the year that lands you in the hospital. Or the pressure ulcer that takes you off your wheelchair for six weeks or months. What does not happen is the slow, almost invisible cardiovascular decline that, twenty years on, becomes the leading cause of death in people with chronic spinal cord injury.
That is a quieter argument than "you will build muscle." It is also the argument that actually matters most over the long horizon, and it is the one that is sometimes poorly served on the open web. I want to present the evidence for preventing so-called secondary complications, including where the evidence ends.
The long tail of spinal cord injury
The hardest thing to grasp in the first year after a spinal cord injury, often, is that the injury itself is not what most defines the next thirty years. It's the complications that can arise from the injury.
The largest prospective cohort study to look at the question, by DeJong and colleagues in 2013, found that more than one in three people with a new traumatic spinal cord injury were rehospitalised at least once in the twelve months after rehabilitation discharge. The three most common reasons were genitourinary (mostly urinary tract infections), respiratory (mostly pneumonia), and skin (mostly pressure ulcers). A 2016 Australian population study by Gabbe and Nunn reported a similar pattern over two years, with urological conditions and pressure injuries leading the readmission list. The picture from the Czech, Turkish, and Dutch cohorts is essentially the same.
The leading drivers of rehospitalisation and quality-of-life loss in chronic SCI are not the injury itself, nor the loss of voluntary movement.
They are infections, skin breakdowns (pressure ulcers), and the long, slow cardiovascular drift that Bauman and Spungen documented over two decades at the James J. Peters VA, and that Weaver and Krassioukov summarised plainly: cardiovascular dysfunction is currently a leading cause of death among those with SCI.
That is the long tail. Anything that meaningfully bends those curves matters more, over a lifetime, than any visible muscle change.
What FES cycling does is well established
The cardiovascular case for FES cycling is the most evidence-based of the four secondary-outcome stories, and it is the right place to start.
The 2021 systematic review by van der Scheer and colleagues pooled 92 studies and 999 adults with SCI. It is the foundation document of the international FES cycling clinical practice guidelines. The review rates the evidence for muscle health benefits as High certainty and for power output and aerobic fitness gains as Low certainty (low because of small sample sizes and design weaknesses, not because the direction of effect is in doubt). Across the included studies, FES cycling consistently improves peak power and peak oxygen uptake during cycling tests, and the metabolic profile shifts in the right direction.
That matters because the metabolic profile is exactly what is broken in chronic SCI. Bauman and Spungen showed in 2001, and the 2015 lipoprotein-particle work by La Fountaine confirmed in finer detail, that the cardiovascular risk picture in chronic SCI is driven by insulin resistance, low HDL particle quality, and abdominal adiposity, not by elevated LDL. Those are precisely the parameters that respond to regular aerobic activity in able-bodied people. FES cycling provides one of the few ways to elicit a meaningful aerobic stimulus in the large lower-limb muscle groups for someone who cannot walk or run.
We have written about the cardiovascular mechanism in more detail in an earlier piece on FES cycling and cardiovascular fitness following a spinal cord injury. For the purposes of this article, the headline is simple: FES cycling addresses the modifiable risk pathway that drives long-term cardiovascular mortality after SCI. It does not make that mortality go away, but it works on the right thing.
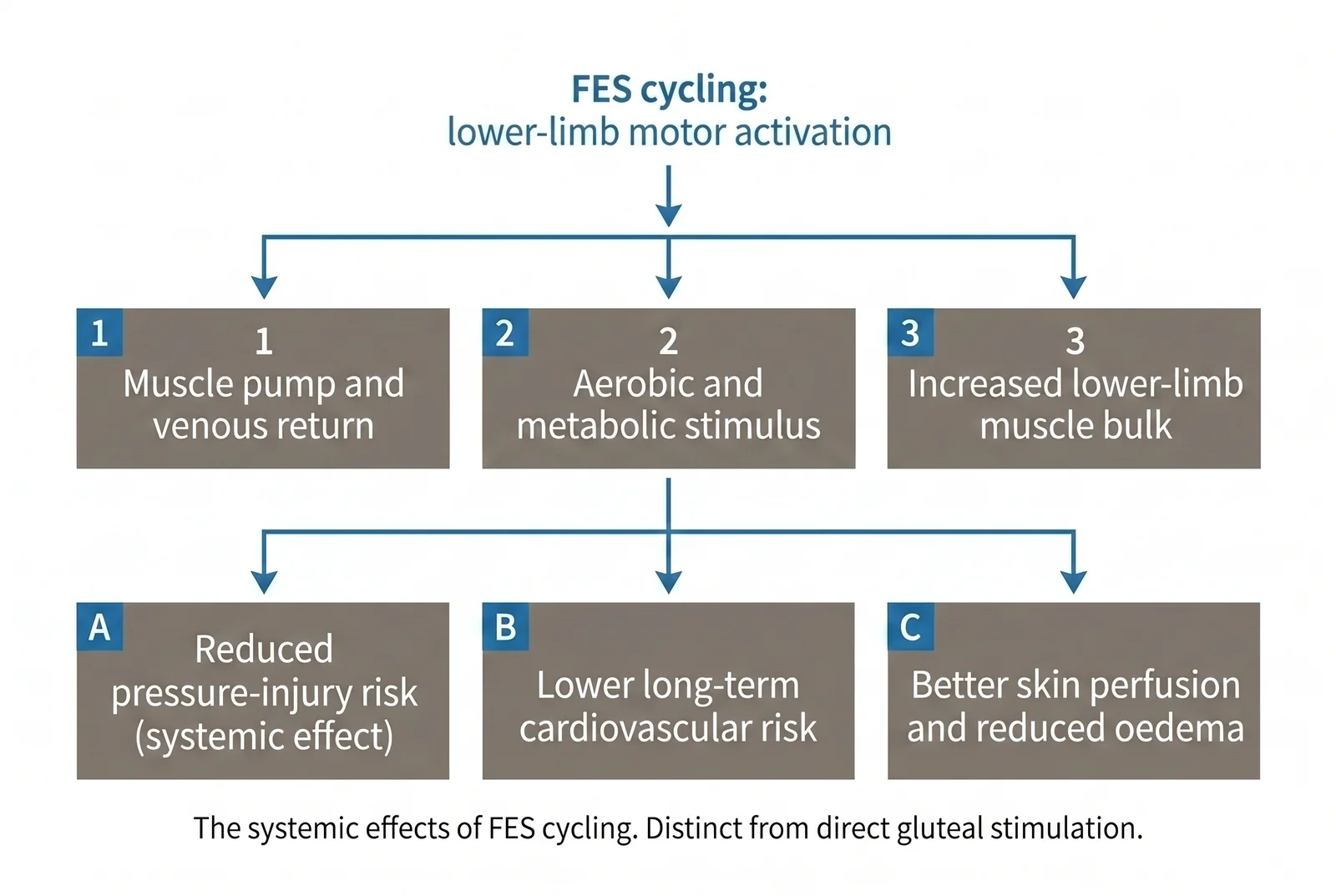
The bladder and bowel case is mechanistically plausible, evidence thin though
The temptation in this kind of article is to lean more heavily on the most positive aspects of the data. We have had clients report that FES cycling has improved their bladder and bowel management, but is there evidence to support this more generally?
For urinary tract infections specifically, I can find no adequately powered randomised trial showing that FES cycling reduces UTI frequency in chronic SCI.
The 2021 van der Scheer systematic review, which is the broadest, explicitly notes that secondary outcomes, such as UTI rates, are not adequately powered across the 92 studies it reviewed. A six-month FES cycling pilot published by Sanna and colleagues in 2025 followed five participants and found no significant change in bowel dysfunction, spasticity, or pain. The strongest evidence for behavioural reduction in UTI rate in SCI comes from a small Cardenas trial of an educational programme, not from any cycling intervention.
The mechanistic case is real enough.
Regular lower-limb activity improves venous return and reduces pelvic venous congestion. Structured exercise tends to anchor the broader self-management discipline (hydration, catheter routine, skin checks) that long-term users develop. Many of my long-term clients tell me they have fewer infections than they used to. I believe them. But "many of my clients tell me" is not population-level evidence.
The bowel picture is similar. Korsten and colleagues showed in 2004 that abdominal wall stimulation (a different intervention) measurably reduced bowel care time and improved colonic motility. That establishes the mechanism: contraction of the abdominal wall has a real effect on defecatory function.
During FES cycling, the recumbent position and the rhythmic contraction of large lower-limb muscles do produce fluctuations in intra-abdominal pressure, which is biologically reasonable as a route to improved bowel transit. But no FES cycling trial has been adequately powered to detect differences in bowel programme length or constipation severity in SCI.
The summary is this: the bladder and bowel benefits that long-term FES cycling users report are mechanistically plausible, biologically reasonable, and not yet demonstrated in trials that would convince a sceptical clinician. Worth tracking. Not yet worth claiming. If a client asks me for a method of using electrical stimulation for bowel care, then I would now use Stim2Go to produce a custom programme for that purpose, not necessarily linked with cycling.
The pressure injury case: drawing a line carefully
This is the section where the evidence picture is richest, and also where it is most often misrepresented. The distinction I want to draw is between FES cycling and direct gluteal electrical stimulation. They are different interventions, and they have different evidence bases.
Direct gluteal stimulation, typically through large surface electrodes targeted specifically at the gluteus maximus, has been shown by Bogie, Liu, and others to increase gluteal muscle thickness by around 50 per cent with regular use, to reduce peak interface pressure under the ischial tuberosities by 20 to 30 per cent, and to roughly double sitting tolerance in people who use it consistently. The 2019 international pressure injury guidelines from EPUAP, NPIAP, and PPPIA provide a conditional recommendation to consider electrical stimulation as an adjunct to standard care for treating recalcitrant Stage III and IV pressure ulcers in adults with SCI. That recommendation is for direct stimulation of the affected tissue, not for whole-body cycling.
I recently wrote on LinkedIn about attending the pressure ulcer summit at Stoke Mandeville, which reinforced the need for prevention. There are many risk factors for pressure ulcers. I'm not going to rewrite that article here except to say that electrical stimulation remains one of the best strategies for preserving tissue bulk and health. In the worst case, when ulcers develop, it can also be one of the best ways of augmenting healing.
FES cycling does something different. It produces systemic effects: increased lower-limb muscle bulk, improved circulation, reduced peripheral oedema, better cardiovascular fitness, and over time a leaner body composition. Each of those is biologically reasonable as a contributor to lower pressure-injury risk. None of them has been demonstrated in a trial powered to detect a change in pressure-injury incidence over the relevant time horizon. We certainly encourage individuals to include gluteal muscle stimulation in their regimen. It can be slightly more awkward to attach electrodes to leg muscles than to other muscles, but certainly worthwhile.
In practice, that means a thoughtful pressure-care strategy might use both. The cycling does the systemic work. Direct stimulation, where indicated, does the local work. The tissue-protection foundations (a properly specified cushion, a posture and positioning plan, the kind of heel offloading we have written about extensively in our pressure ulcer and PRAFO articles) sit underneath all of it. NICE CG179, the UK guideline on pressure ulcer prevention and management, does not currently recommend electrical stimulation for routine pressure-injury prevention it remains a treatment adjunct in specific clinical contexts.
I am willing to say, based on years of clinical observation, that long-term FES cycling users in our practice tend to have fewer skin events than we would expect for their level of injury. That is consistent with the systemic mechanism story. It is not the same as a randomised demonstration, though.
What to track over twelve months
If you are using FES cycling primarily for long-term secondary complication prevention, the wrong way to evaluate it is on a weekly or monthly basis. The events you are trying to influence are infrequent and noisy at short timescales. Twelve months is the right window.
Here is a defensible tracking plan, drawn from validated outcome instruments where they exist.
- UTI frequency. Date-and-symptom log, antibiotic episodes. The International SCI UTI Basic Data Set (Goetz, Cardenas et al., 2013) provides the appropriate field structure if you want to be rigorous.
- Bowel programme length and continence. The International SCI Bowel Function Basic Data Set, version 2.0 (Krogh, Emmanuel et al. 2017) records average defecation time, frequency, incontinence episodes, and use of stimulation.
- Sitting tolerance. Self-reported maximum tolerable sitting time before discomfort or pressure-relief need. This is the patient-meaningful outcome that Bogie used.
- Skin status. Monthly photo log of high-risk areas: sacrum, ischial tuberosities, heels, trochanters. Stage and document any breakdown.
- Lower-limb circumference and oedema. Standardised tape measurement at the calf and ankle, taken at the same time of day.
- Resting blood pressure (monthly home reading) and orthostatic check (quarterly).
- Annual GP review, including lipid panel and HbA1c or fasting glucose. This is where the Bauman metabolic-syndrome story becomes a tracked, modifiable parameter.
- Transfer ease and functional independence. A simple self-rated 1-to-10 scale captures changes that matter to the user.
- Cycling-specific outputs. Session duration, peak power, and perceived exertion. Useful for adherence and progression.
Pick three or four of these that you genuinely care about. Track them monthly for a year. The discipline matters more than the choice of metric.
Why is this case rarely made?
One reason is straightforward: muscle changes photograph well, and "what did not happen" does not. There is no before-and-after image of the UTI or pressure ulcer you did not have.
Another reason is structural. The trials needed to show a meaningful reduction in UTI rate or pressure injury incidence in chronic SCI are large, multi-centre, expensive, and slow. The trials that get funded and published are the ones that fit a doctoral timeline, which means they are usually small and powered for fitness or muscle outcomes. The long-game case is not in the literature in the form a clinician would want, because the literature is not designed to answer it that way.
The third reason is harder to name. The argument is not exciting. It does not promise transformation. It promises, more or less, fewer bad weeks over a long life.
Some limitations
The argument I have made here has clear limits.
Most FES cycling RCTs are not powered to detect secondary complication outcomes. Sample sizes in the relevant studies are typically below 20, sometimes below 10. There is no published trial demonstrating that FES cycling alters EDSS, life expectancy, or pressure-injury incidence at the population level. People who maintain FES cycling for ten years are self-selected, and some of the benefits attributed to the cycling may reflect the broader self-management discipline these long-term users develop. None of that invalidates the mechanism story, but it should keep the language careful.
A practical next step
If you are at the start of an SCI rehabilitation pathway, or supporting a family member who is, the most useful thing you can do is plan around the next few years, not the next twenty minutes. The technology you put in place now is a long-game investment, and it is worth choosing it that way.
That is what an assessment is for, in our case. It is a conversation about the level and completeness of injury, the home setup, the family and care support, the goals that matter most, and what realistic outcomes look like at one year, five years, and twenty years. Sometimes FES cycling is the central piece. Sometimes it sits alongside other things. Sometimes the right answer is "not yet, the priority is something else." All three are useful answers when they are the honest one.
If you would like to talk through your situation, please get in touch. The fitness story is the easy one to tell. The long-game story is the one that matters.
Further Reading
Bauman WA, Spungen AM. Carbohydrate and lipid metabolism in chronic spinal cord injury. Journal of Spinal Cord Medicine. 2001. https://doi.org/10.1080/10790268.2001.11753584
Bauman WA et al. 31st G. Heiner Sell lectureship: secondary medical consequences of spinal cord injury. Topics in Spinal Cord Injury Rehabilitation. 2012. https://doi.org/10.1310/sci1804-354
Bogie KM, Wang X, Triolo RJ. Long-term prevention of pressure ulcers in high-risk patients: a single case study of the use of gluteal neuromuscular electric stimulation. Archives of Physical Medicine and Rehabilitation. 2006. https://doi.org/10.1016/j.apmr.2005.11.020
Cardenas DD, Hoffman JM, Kelly E, Mayo ME. Impact of a urinary tract infection educational program in persons with spinal cord injury. Journal of Spinal Cord Medicine. 2004. https://doi.org/10.1080/10790268.2004.11753730
DeJong G et al. Rehospitalization in the first year of traumatic spinal cord injury after discharge from medical rehabilitation. Archives of Physical Medicine and Rehabilitation. 2013. https://doi.org/10.1016/j.apmr.2012.10.037
EPUAP/NPIAP/PPPIA. Prevention and Treatment of Pressure Ulcers/Injuries: International Clinical Practice Guideline (3rd edition). 2019. https://www.internationalguideline.com/2019
Gabbe BJ, Nunn A. Profile and costs of secondary conditions resulting in emergency department presentations and readmission to hospital following traumatic spinal cord injury. Injury. 2016. https://doi.org/10.1016/j.injury.2016.06.012
Goetz LL, Cardenas DD et al. International Spinal Cord Injury Urinary Tract Infection Basic Data Set. Spinal Cord. 2013. https://doi.org/10.1038/sc.2013.72
Korsten MA et al. Difficulty with evacuation after spinal cord injury: colonic motility during sleep and effects of abdominal wall stimulation. Journal of Rehabilitation Research and Development. 2004. https://doi.org/10.1682/jrrd.2004.01.0095
Krogh K, Emmanuel A et al. International spinal cord injury bowel function basic data set (Version 2.0). Spinal Cord. 2017. https://doi.org/10.1038/sc.2016.189
La Fountaine MF et al. Lipoprotein heterogeneity in persons with spinal cord injury. Lipids in Health and Disease. 2015. https://doi.org/10.1186/s12944-015-0084-4
Liu LQ, Ferguson-Pell M. Pressure changes under the ischial tuberosities during gluteal neuromuscular stimulation in spinal cord injury. Archives of Physical Medicine and Rehabilitation. 2014. https://doi.org/10.1016/j.apmr.2014.10.008
Martin Ginis KA, van der Scheer JW et al. Evidence-based scientific exercise guidelines for adults with spinal cord injury. Spinal Cord. 2018. https://doi.org/10.1038/s41393-017-0017-3
NICE. Pressure ulcers: prevention and management. CG179. 2014 (last reviewed 2018). https://www.nice.org.uk/guidance/cg179
NICE. RT300 for spinal cord injury rehabilitation. MIB169. 2018. https://www.nice.org.uk/advice/mib169
Sanna N et al. Evaluating the health and fitness benefits of a 6-month FES-cycling program on a recumbent trike for individuals with motor complete SCI: a pilot study. Journal of NeuroEngineering and Rehabilitation. 2025. https://doi.org/10.1186/s12984-025-01585-0
Smit CAJ, de Groot S, Stolwijk-Swuste JM, Janssen TWJ. Effects of electrical stimulation on risk factors for developing pressure ulcers in people with a spinal cord injury: a focused review. American Journal of Physical Medicine and Rehabilitation. 2016. https://doi.org/10.1097/PHM.0000000000000501
van der Scheer JW, Goosey-Tolfrey VL, Valentino SE, Davis GM, Ho CH. Functional electrical stimulation cycling exercise after spinal cord injury: a systematic review of health and fitness-related outcomes. Journal of NeuroEngineering and Rehabilitation. 2021. https://doi.org/10.1186/s12984-021-00882-8
Weaver LC, Fleming JC, Mathias CJ, Krassioukov AV. Disordered cardiovascular control after spinal cord injury. Handbook of Clinical Neurology. 2012. https://doi.org/10.1016/B978-0-444-52137-8.00013-9

